|
the possible magnetic quantum numbers are -l, -l+1.0., l-1, l.the number of magnetic quantum numbers m l=2l+1.Magnetic quantum number determines the energy shift of the atomic orbitals after it is exposed to a magnetic field. Magnetic quantum number is related to the angular momentum. Magnetic quantum number is not related to the energy of each orbital but the probability of the electron. Magnetic quantum number, m l, describes the quantum state designated m. There can be 3 p orbitals with the same energy with in a subshell (p x, p y, p z). The possible number of electrons in each subshell is described by 2 l+1.ĭegeneracy orbitals are orbitals that have the same energy. The possible angular momentum quantum numbers are - l-1.0. l=0 refers to the s orbital, l=1 refers to the p orbital, and l=2 for d orbitals, etc. Different shells have different energy levels.Īngular momentum quantum number, l, refer to the subshells of principle quantum numbers. 
The number of electrons that can be placed in a shell is described by the equation 2n 2. Principle quantum number, n, indicates what shell the electron belongs to. It is often helpful for students to think of quantum states as analogous to addresses (country, city, street, house number, name of the person, etc.) Each electron can only have one quantum state. 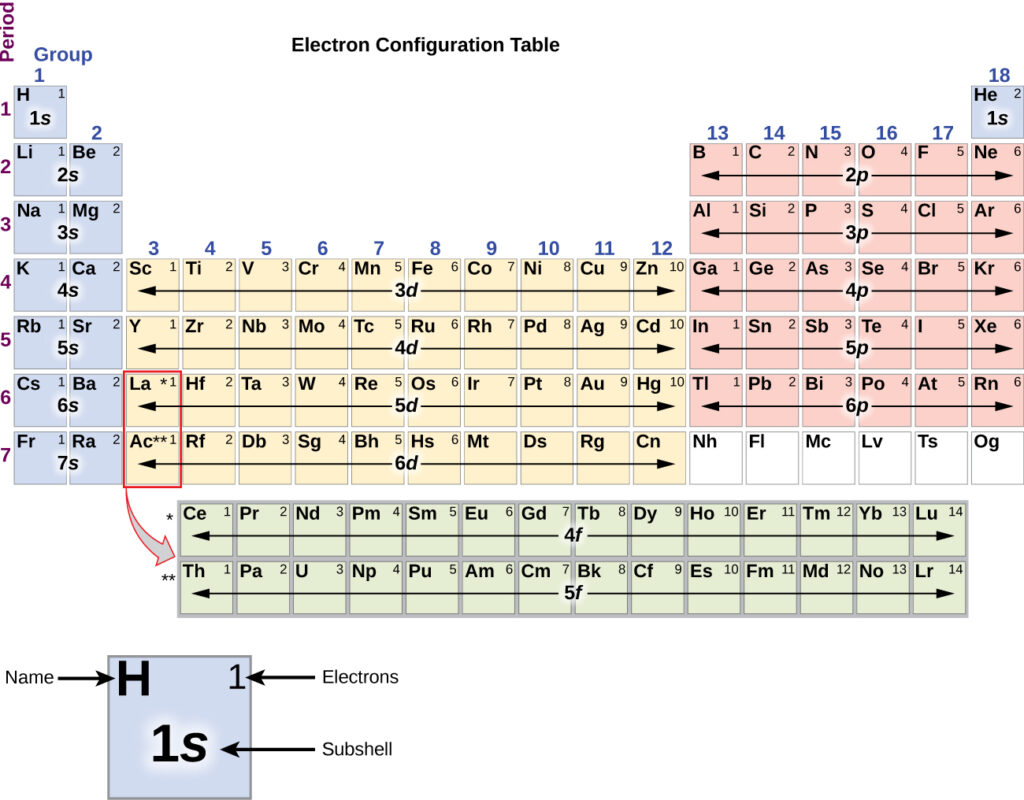
Orbitals are placed in their shells and subshells, which are governed by four quantum numbers: n, l, m l, m s. This article is part of the ChemHelp Tutoring WikiĮlectron configuration is a model based on quantum chemistry to show how electrons will be arranged in different orbitals.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
